What is the PKA Micro-Needle’s market potential?
PKA SoftTouch Corp.’s growth is closely linked to the continued growth of injectable drug applications and markets. The medical device market offers investors unique exposure to the overall life science space, especially in an era of fast-growing tech advancements in healthcare.
The market calls for an alternative to the commonplace syringe, the dominant injection device. Our technology is easily adaptable to a wide variety of uses in both human and animal markets, including, but not limited to diabetes, tuberculosis screening, heparin, pediatrics, obesity drugs, treatment for opioid overdoses and pain medications. The device also has high potential use in vaccination markets internationally to control the spread of viruses, such as the Coronavirus, the Ebola virus, and other life-threatening diseases such as Diptheria, Malaria, Cholera, and Typhoid.
Injectable Global Drug Market Potential
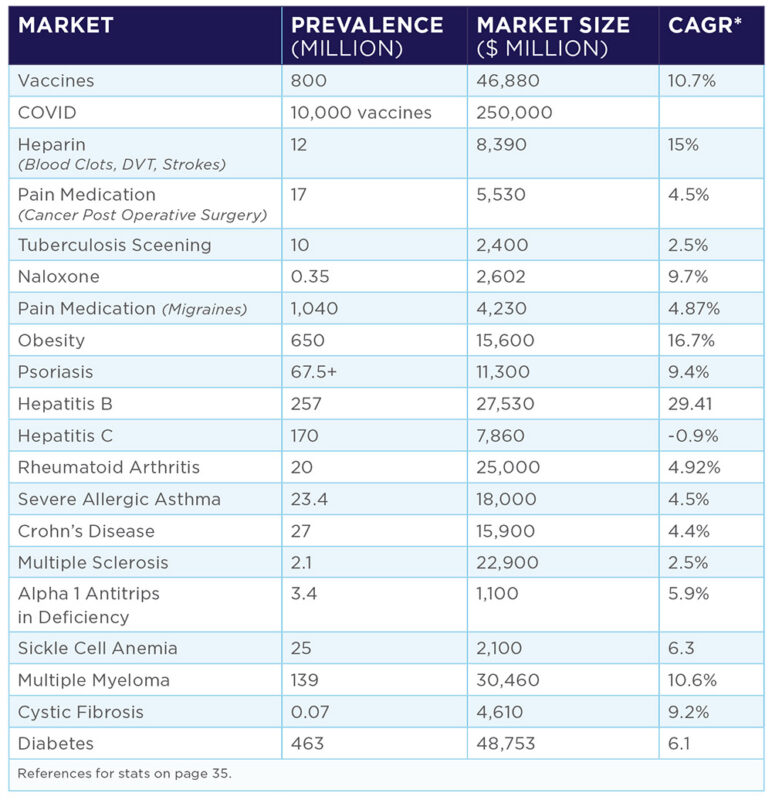
COVID-19 PANDEMIC:
*National Post June 12 2020 **National Post July 14 2020
BY 2024:
*Mordon Intelligence **IDF Diabetes Atlas 9th Edition 2019 ***Zion Market research
Market Potential by 2024
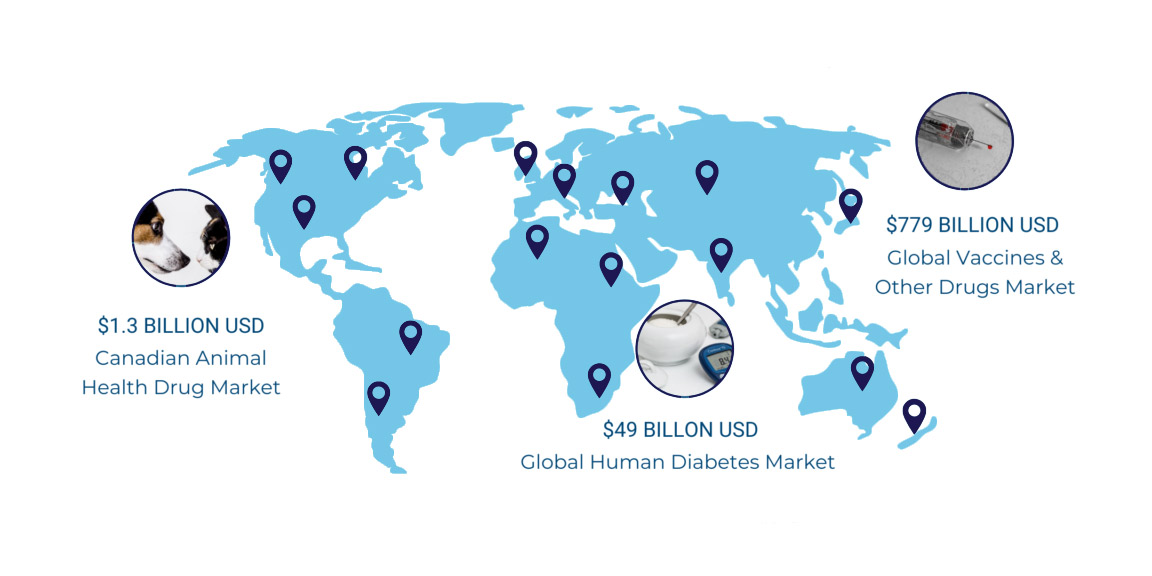
The PKA SoftTouch Micro-Needle is Patented in 23 Countries – with more patents pending.
NORTH AMERICAN INSULIN MARKET POTENTIAL BY 2024
With global diabetes continuing to grow at an alarming rate, insulin represents the largest injectable drug market worldwide. In fact, more healthcare dollars were spent on diabetes treatment in the U.S than on any other illness in 2019. Approximately 463 million adults are living with diabetes and by 2045 this will rise to 700 million. By 2060 it is anticipated that one in three adults in the U.S. will be diabetic (68 million people). Diagnosed diabetes in Canada and the U.S. is expected to increase 171 per cent from 2014 to 2060*.
This growth rate reflects both the increased prevalence among the older demographic as well as the increased prevalence of obesity and related diabetes risk factors among youth. In the context of this growth in the patient population, diabetes disease management and technology must evolve at a greater rate. PKA aims to be part of the evolution.
The veterinary and animal health industry offers a high potential market with companion animals posing fewer regulatory hurdles and therefore an attractive avenue for market entry, followed by humans. Hence, the initial geographical market focus for PKA is the United States and Canada.
*International Diabetes Federation, Diabetes Atlas, 5th Ed. :The Global Burden **Canada (2018) & USA (2017) Sources: Canadian Animal Health Institute, American Veterinary Medical Association, Statista. com *** Canada (2017) & USA (2017) Sources: Healthline, Government of Canada
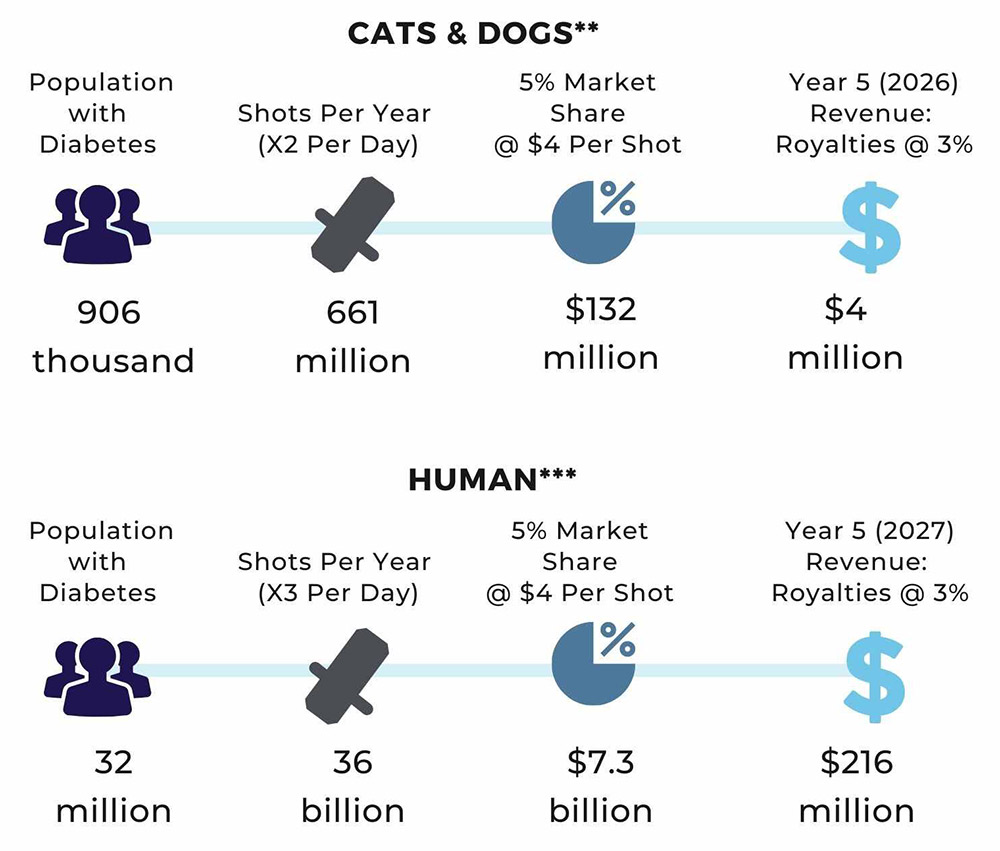
ONE STEP AWAY FROM COMMERCIALIZATION
Notably, we are well on our way through the drug approval pathway process.
We have refined our technology and our Generation VI Micro-Needle, our final and perfected device, was finalized in 2018. It has passed all testing with 100 per cent reliability. To receive regulatory approvals, we now must confirm efficacy and safety. Our next step is to complete veterinary clinical trials, which will be conducted in late May 2021 at the University of Guelph, Ontario.
Part 1 of this two-step process is using easily modified 3-D technology for our medical device. The second and final part of the veterinary trials takes place in December 2021. By then we will have tweaked and refined our device technology by curating moulds to the point of perfection and complete reliability and lower costs.
For further details on this investment offering and how to invest, please click below.
LEARN MOREIf you have additional questions, please contacts us.


